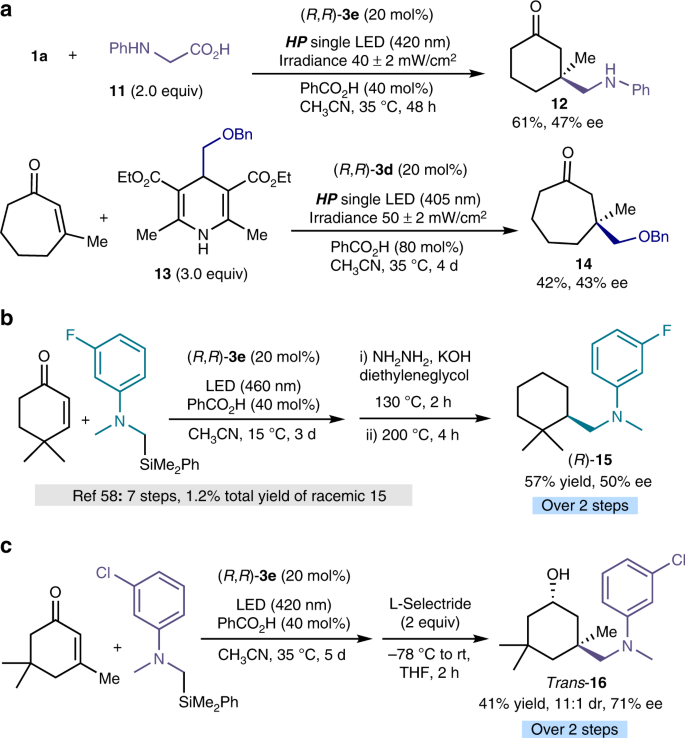
Enantioselective radical conjugate additions driven by a photoactive intramolecular iminium-ion-based EDA complex | Nature Communications

Synthetic Methods Driven by the Photoactivity of Electron Donor–Acceptor Complexes | Journal of the American Chemical Society
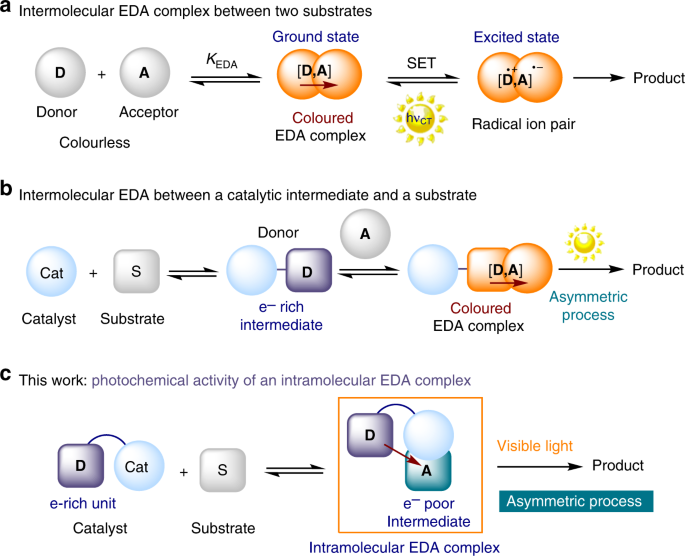
Enantioselective radical conjugate additions driven by a photoactive intramolecular iminium-ion-based EDA complex | Nature Communications

Synthetic Methods Driven by the Photoactivity of Electron Donor–Acceptor Complexes | Journal of the American Chemical Society

Recent advances in catalyst-free photochemical reactions via electron-donor-acceptor (EDA) complex process - ScienceDirect

Overcoming Back Electron Transfer Facilitates the Implementation of Electron Donor-Acceptor Complexes in Catalysis | Catalysis | ChemRxiv | Cambridge Open Engage

Synthetic Methods Driven by the Photoactivity of Electron Donor–Acceptor Complexes | Journal of the American Chemical Society
Design and Implementation of a Catalytic Electron Donor-Acceptor Complex Platform for Radical Trifluoromethylation and Alkylatio

Aerobic Oxidative EDA Catalysis: Synthesis of Tetrahydroquinolines Using an Organocatalytic EDA Active Acceptor - ScienceDirect

J. Am. Chem. Soc. on Twitter: "Modular class of organic catalysts that as donors can readily form photoactive electron donor–acceptor (EDA) complexes with a variety of radical precursors @MelchiorreGroup @Melchiorre_P https://t.co/yFsLDrlflE" /
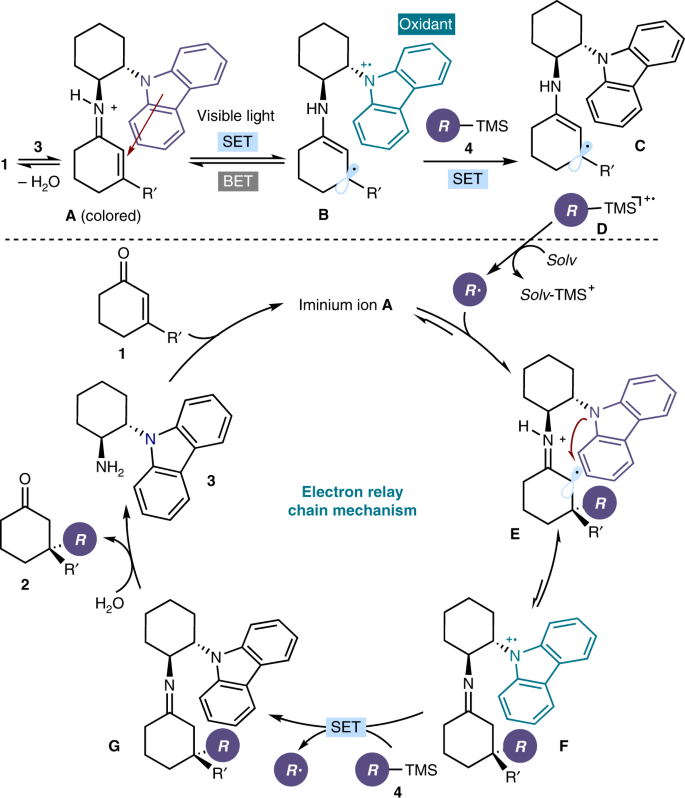
Enantioselective radical conjugate additions driven by a photoactive intramolecular iminium-ion-based EDA complex | Nature Communications
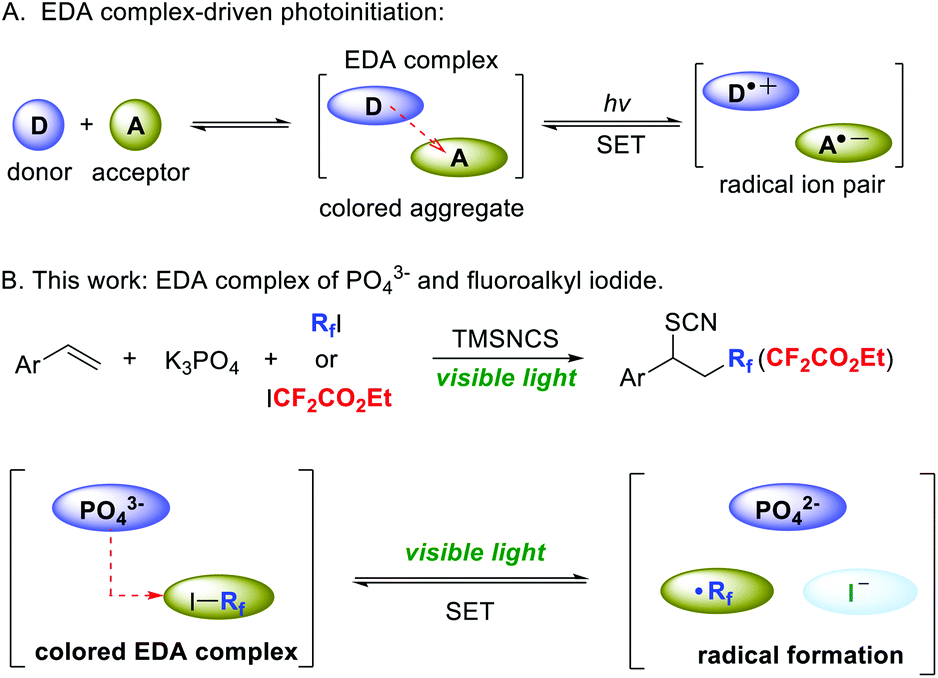
Visible light-driven fluoroalkylthiocyanation of alkenes via electron donor–acceptor complexes - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00126D
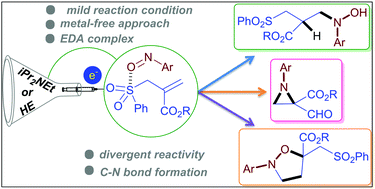
EDA complex directed N-centred radical generation from nitrosoarenes: a divergent synthetic approach - Chemical Communications (RSC Publishing)
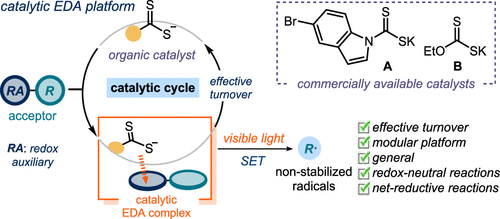
A General Organocatalytic System for Electron Donor–Acceptor Complex Photoactivation and Its Use in Radical Processes,Journal of the American Chemical Society - X-MOL
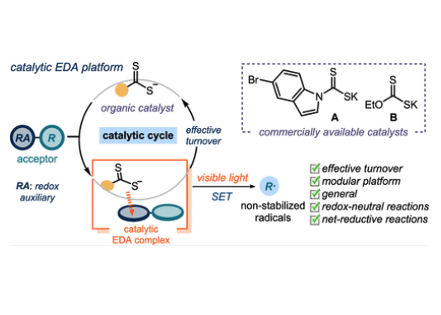
A General Organocatalytic System for Electron Donor−Acceptor Complex Photoactivation and Its Use in Radical Processes
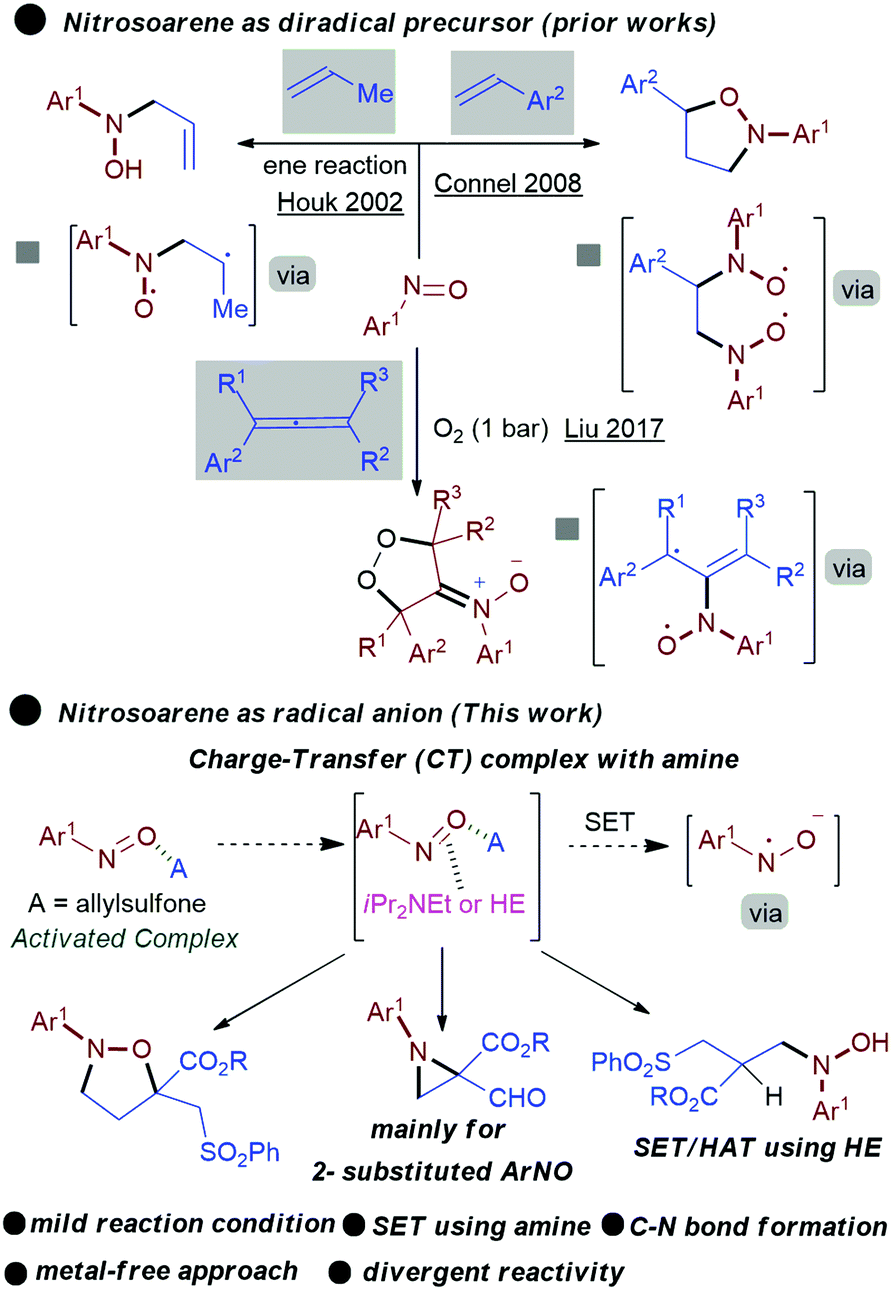
EDA complex directed N-centred radical generation from nitrosoarenes: a divergent synthetic approach - Chemical Communications (RSC Publishing) DOI:10.1039/C9CC07277B

Synthetic Methods Driven by the Photoactivity of Electron Donor–Acceptor Complexes | Journal of the American Chemical Society


