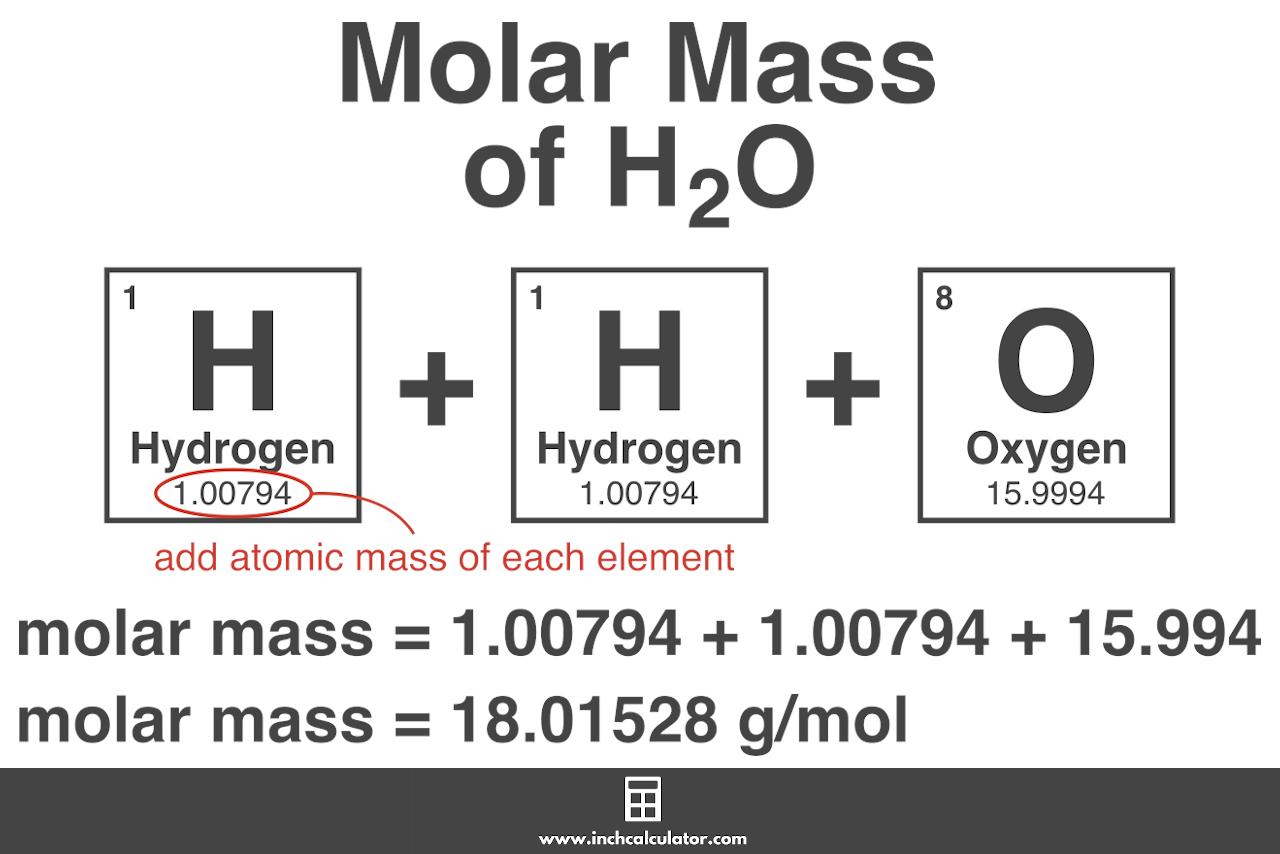
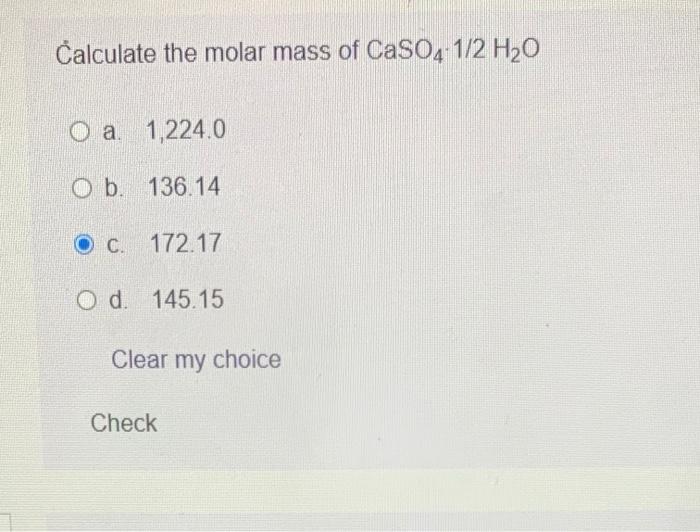
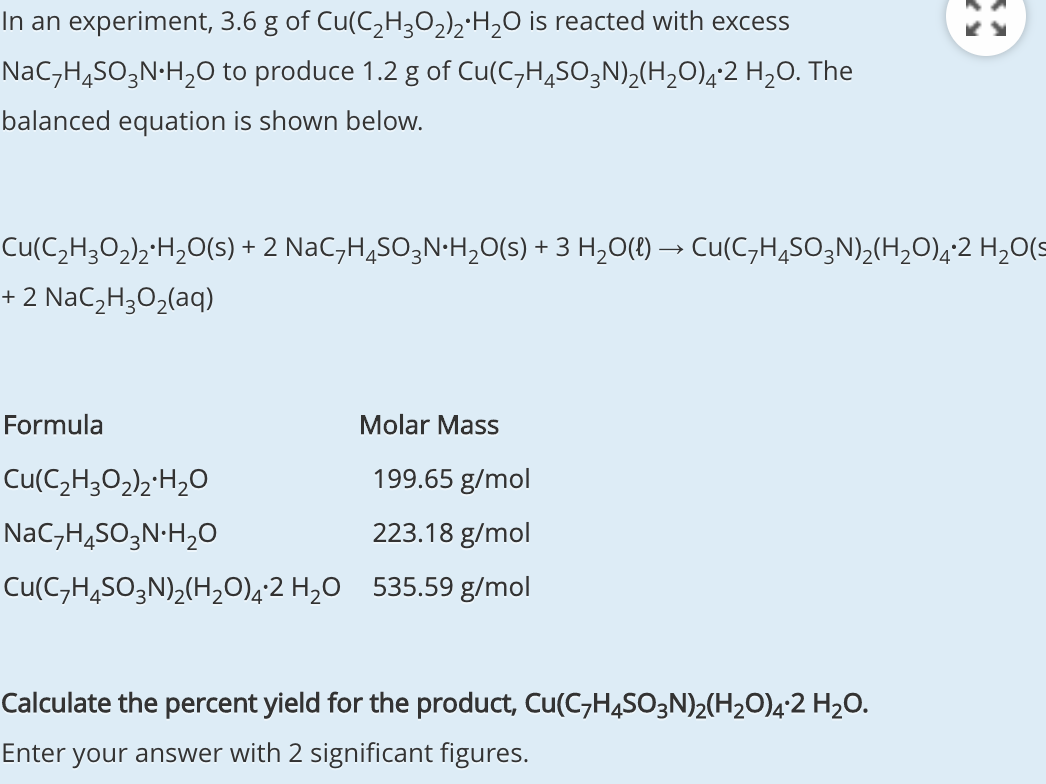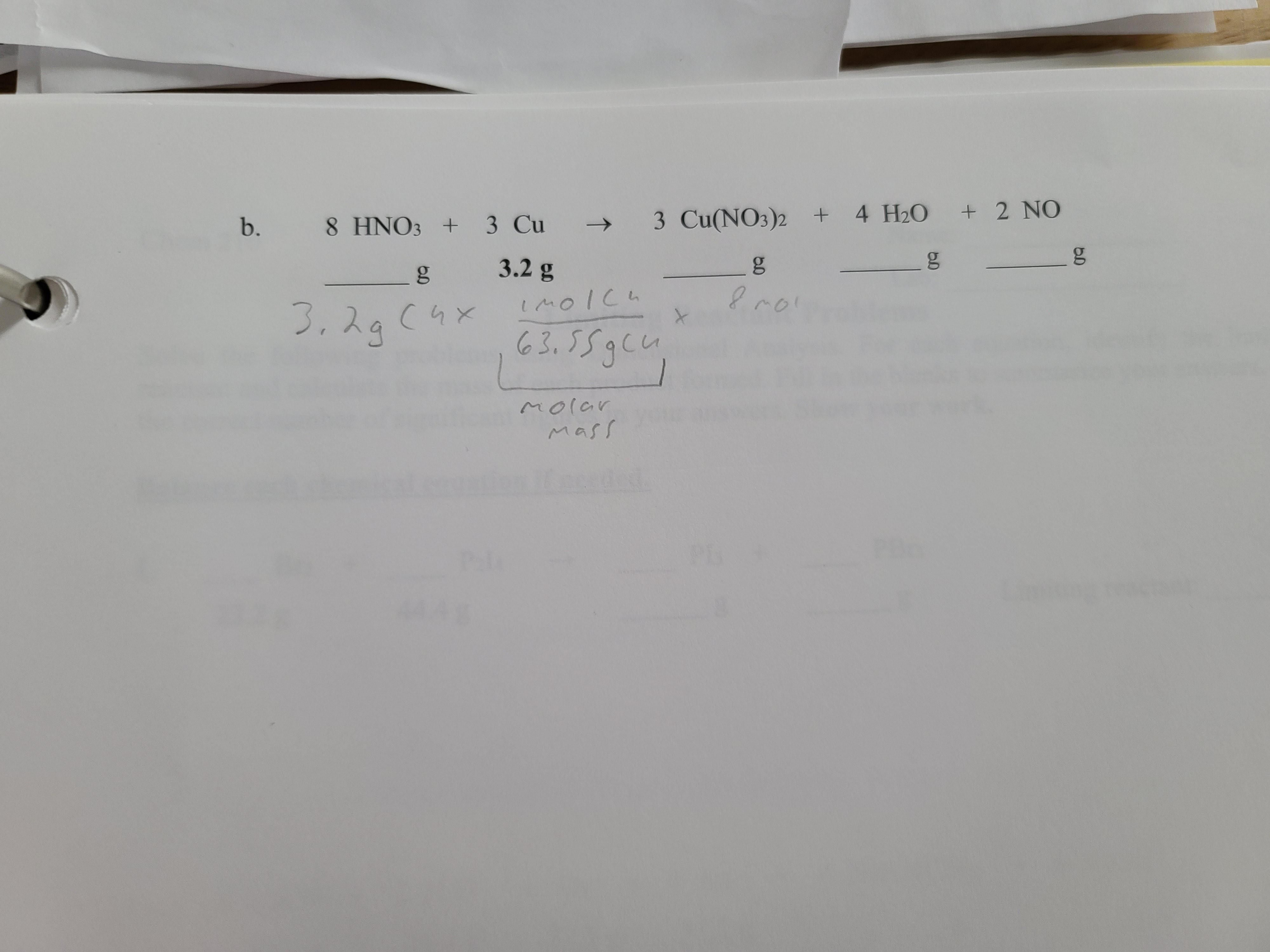SOLVED: What mass in grams of the dihydrate H2C2O4 • 2 H2O (molar mass = 126.0 g/mol) is needed in order to deliver 16.4 grams of H2C2O4 (molar mass = 90.0) to

Calculate Calculate the molar mass of H2O - Chemistry - Some Basic Concepts of Chemistry - 12618097 | Meritnation.com

a) Calculate the relative molecular mass of water `(H_(2)O)`. (b) Calculate the molecular mass ... - YouTube

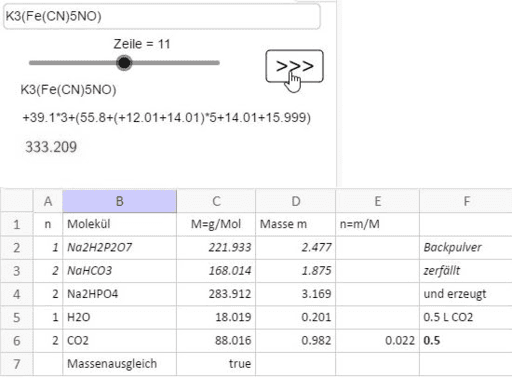
Molar mass of co(NO3)2 6H2O is 290grams given mass is 30grams in 4 3litres of solution calculate molarity - Chemistry - - 13950035 | Meritnation.com
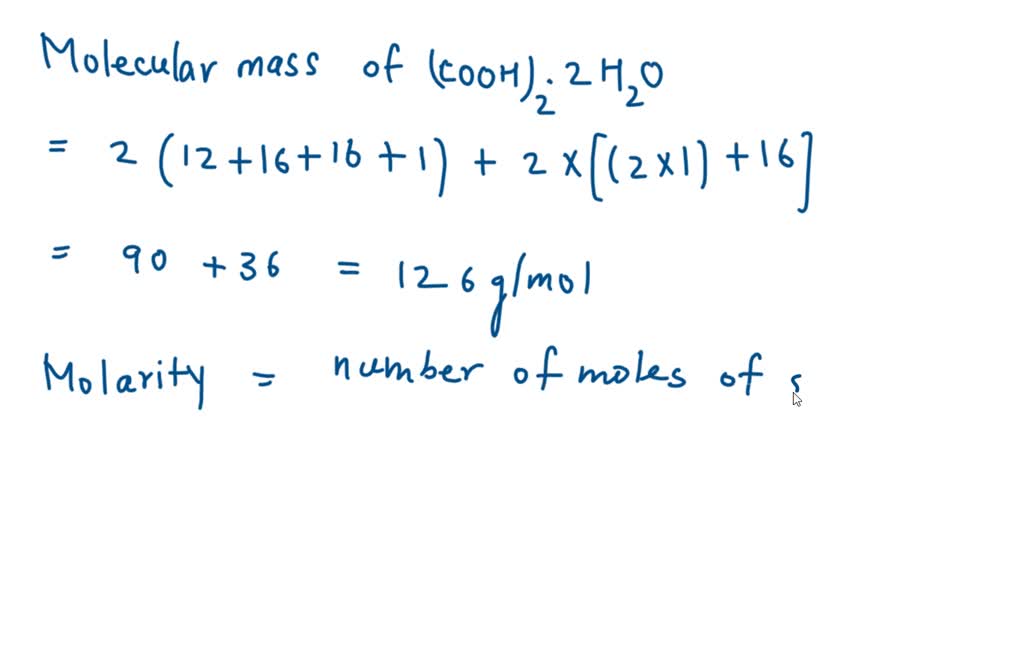
SOLVED: Amount of oxalic acid (COOH)2.2H2O, in grams required to prepare 200mL of 0.5 M oxalic acid solution is