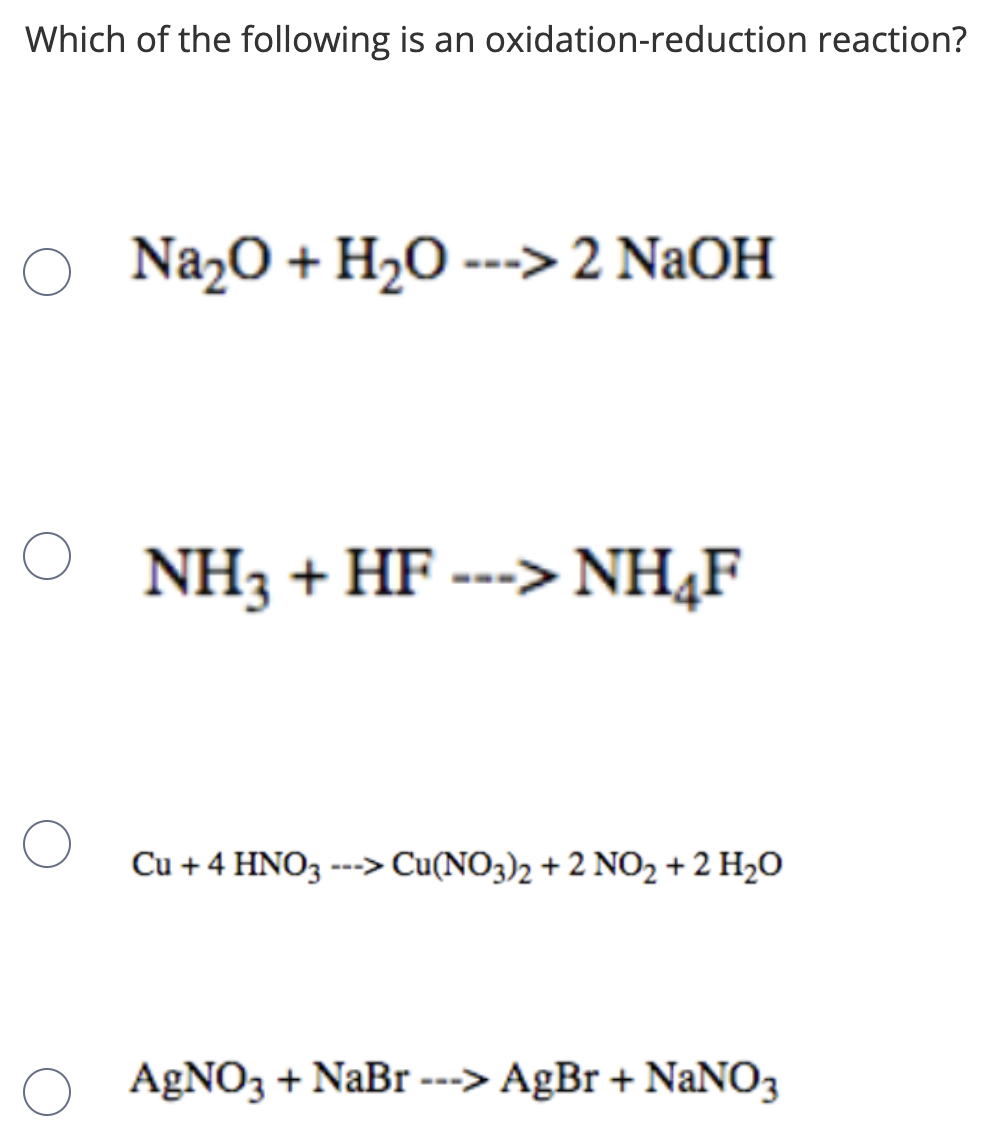Sodium Oxide (Na2O) - Structure, Physical Properties, Chemical Properties and Uses with FAQs of Sodium Oxide

L28 Piston for Nissan Engine Na20/E13/H20/H51/K25/Td25/Td27 Piston - China Suzuki Piston and Td27 Piston

2. Дописать уравнения реакций, расставить коэффициенты и указатьтип химической реакции1)CO2 - Школьные Знания.com

Auto Parts carburador (NA20) OEM: 16010-J0502 Nissan H20 Junior - China Autopartes, CARBURADOR (NA20)

SOLVED: Given the following equation: Na2O + H20 - 2 NaOH How many grams of Na20 are required to produce 1.60 x 102 grams of NaOH? g

Bulletin - United States National Museum. Science. THE MINERALS OF IDAHO 375 scent marcasite from the Garfield tunnel. This was analyzed with the following results: Analysis and ratios of clay from Garfield tunnel (E. V. Shannon, analyst) Per cent Ratios ...
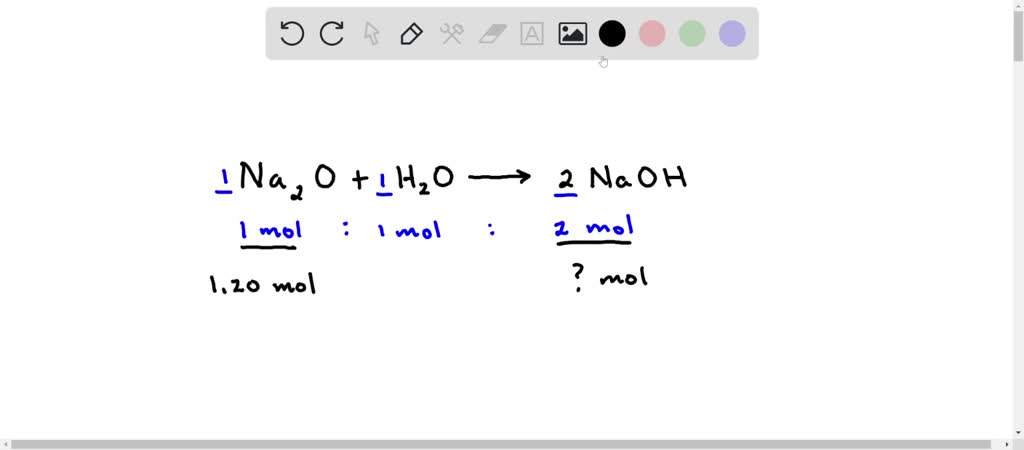
SOLVED: Given the following equation: Na2O + H2O —> 2 NaOH How many moles of NaOH are produced from 1.20 moles of Na2O?