The system Al2O3-P2O5-H2O at temperatures below 200 °C: Experimental data on the stability of variscite and metavariscite AlPO4·2H2O | Semantic Scholar
Complete and balance the following equation (i) P2O5 + H2O → ............ (ii) S + O2 → ................ - Sarthaks eConnect | Largest Online Education Community

One mole of P2O5 undergoes hydrolysis as P2O5 + H2O⟶H3PO4 The normality of the phosphoric acid formed is (The volume of solution is 1 L)
Complete and balance the following equations: 1. NH3 + CaCl2 → 2. NH3 + P2O5 + H2O → - Sarthaks eConnect | Largest Online Education Community

One mole of P2O5 undergoes hydrolysis as P2O5 + H2O⟶H3PO4 The normality of the phosphoric acid formed is (The volume of solution is 1 L)

PDF) Heat capacity and glass transition in P2O5–H2O solutions: support for Mishima's conjecture on solvent water at low temperature | Federico Nores Pondal - Academia.edu
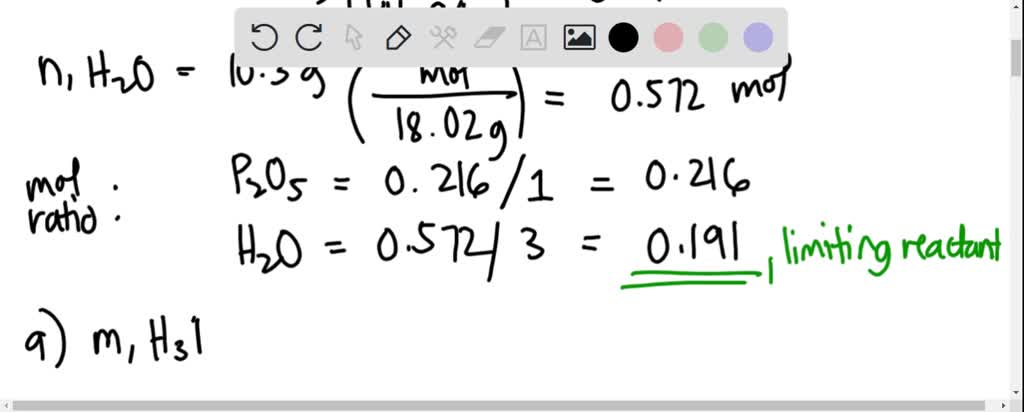
SOLVED: Identify the limiting reactant in the reaction of diphosphorus pentoxide and water to form H3PO4, if 27.4 g of P2O5 and 14.2 g of H2O are combined. Determine the amount (in
![PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e9e458615e33c40d94b80a3189efa26d438c8a39/4-Figure2-1.png)
PDF] Heat capacity and glass transition in P2O5-H2O solutions: support for Mishima's conjecture on solvent water at low temperature. | Semantic Scholar

Total Ni, exchangeable ions, available P2O5, and pH (H2O) in the soils... | Download Scientific Diagram